Microdosing Semaglutide Chart: A Monthly Dose Guide For Josie Patients
A semaglutide microdosing chart that supports comfort, longevity, and gentle month-to-month increases.
GLP-1 microdosing is gaining attention in menopause. Here’s how hormones, metabolism, and dosing intersect.

The Josie Team
Medically Reviewed by


GLP-1 microdosing is increasingly discussed among women navigating perimenopause and menopause, particularly as hormonal changes begin to influence metabolic regulation and medication tolerance. In this article, the term refers to dosing strategies below the initiation levels used in large clinical trials (see Key Definitions below).
Medications such as semaglutide and tirzepatide work through the same biological mechanisms at any age. What changes in midlife is the metabolic environment those medications enter.
During perimenopause and menopause, estrogen and progesterone levels shift. Those hormonal changes influence insulin sensitivity, appetite regulation, body composition, and medication tolerance. Because of that, the same GLP-1 dose can feel different in midlife than it does earlier in life.
Understanding GLP-1 dosing in midlife requires looking at the full metabolic picture. Hormones influence metabolism, metabolism shapes medication response, and medication response affects how different dosing strategies behave over time.
Microdosing is not hormone therapy, and it does not independently protect muscle mass. It is a dosing strategy that must be evaluated within a broader framework of hormonal health, metabolic stability, and lean mass preservation.
And sequence determines outcome.
In standard clinical practice, GLP-1 therapies typically begin at defined initiation doses and gradually increase through titration. Discussions of microdosing often emerge when patients experience difficulty tolerating those starting doses or when clinicians attempt slower dose escalation. In these situations, lower-dose strategies may be explored as a tolerance-focused approach rather than a method intended to maximize metabolic effect.
GLP-1 microdosing refers to using doses below the validated initiation levels studied in large randomized clinical trials of GLP-1 receptor agonists such as semaglutide and tirzepatide.
Dose calibration refers to adjusting GLP-1 exposure relative to an individual’s metabolic response, tolerance profile, and hormonal environment.
The hormone-aware metabolic framework describes a structured sequence for metabolic intervention: stabilizing hormonal conditions first, calibrating GLP-1 dosing second, accelerating metabolic improvement through monitoring and titration, and preserving lean mass throughout the process.
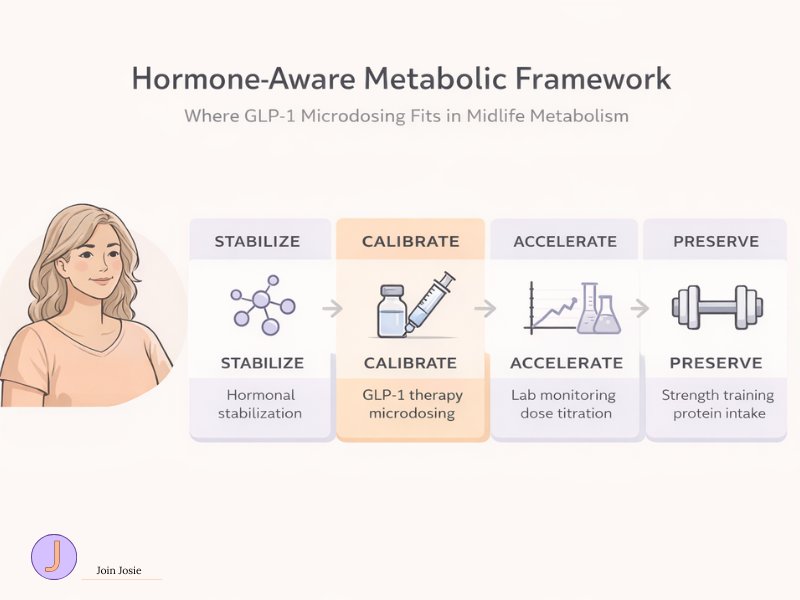
GLP-1 microdosing should be evaluated within a structured order of metabolic care rather than as a stand-alone dosing experiment. When dose strategy is separated from hormonal health, muscle preservation, and metabolic monitoring, short-term appetite reduction may occur without durable metabolic improvement.
For midlife women, the metabolic framework can be understood as a four-phase sequence. The hormone-aware metabolic framework presented here is an educational model designed to organize current knowledge about menopause physiology, metabolic regulation, and GLP-1 therapy. It is not a clinical guideline but a conceptual structure for understanding how these factors interact.
Microdosing belongs in the Calibrate phase. It does not replace Stabilize, and it cannot substitute for Preserve. Dose adjustments cannot compensate for untreated hormonal instability or ongoing muscle loss.
Hormonal status affects insulin sensitivity, and insulin sensitivity strongly influences how a body responds to GLP-1 therapy. When those underlying conditions are stable, medication adjustments are more likely to produce durable metabolic results.
When microdosing is discussed outside this sequence, it becomes a tactic without a strategy.
GLP-1 dose calibration becomes appealing in menopause because several biological shifts occur at the same time. These shifts influence insulin sensitivity, appetite regulation, and body composition.
The growing interest in microdosing is therefore grounded in physiology, not simply trend.
Four interlocking changes define the midlife metabolic environment.
Estrogen decline directly affects insulin sensitivity and fat distribution. As estrogen levels fall, muscle and liver cells respond less efficiently to insulin. The pancreas compensates by releasing more insulin to manage the same glucose load.
Over time this pattern may lead to:
Many women notice these metabolic changes before seeing meaningful changes on the scale.
When insulin resistance increases, circulating insulin levels rise as the body attempts to control blood glucose. Elevated insulin levels encourage fat storage, and increasing fat mass can further worsen insulin resistance over time. This feedback loop can reinforce itself if the underlying metabolic drivers are not addressed.
GLP-1 receptor agonists improve glucose-dependent insulin signaling and help reduce post-meal glucose spikes. That mechanism explains why GLP-1 therapy may support metabolic regulation during midlife.
However, if systemic estrogen deficiency remains unaddressed, insulin resistance may continue to be hormonally driven. For that reason, calibration generally works best after hormonal status has been evaluated.
Menopause often shifts fat storage from subcutaneous areas to visceral abdominal depots, even when total body weight remains stable. Visceral fat is metabolically active and releases inflammatory signals that influence insulin resistance and cardiovascular risk.
This is one reason scale weight alone becomes an unreliable marker of metabolic health in midlife women.
Standard-dose GLP-1 therapy has demonstrated reductions in visceral fat in clinical trials. Microdosing has not been studied at sub-therapeutic levels for this outcome.
Visceral fat contributes to systemic inflammation, and chronic inflammation can worsen insulin resistance. As insulin resistance increases, the body becomes more likely to store additional fat in visceral depots, reinforcing the metabolic cycle.
Calibration strategies often aim to interrupt this cycle, but the strength and reliability of that effect depend on dose levels that have been validated in clinical trials.
Estrogen supports muscle protein synthesis and recovery. As estrogen levels fall, lean mass declines more rapidly, lowering resting metabolic rate and reducing glucose disposal capacity.
This change is often subtle at first. Many women report eating similarly to their 30s while gaining weight more easily in their 40s and 50s. Reduced muscle mass partially explains that shift.
GLP-1 therapy can compound this risk. In the STEP-1 trial published in the New England Journal of Medicine, approximately 38–40% of total weight lost during semaglutide treatment came from lean mass rather than fat. For a midlife woman losing 20 pounds, that could represent roughly eight pounds of muscle if resistance training is not maintained.
Muscle mass plays a major role in metabolic rate, and metabolic rate strongly influences long-term weight stability. When lean mass declines, the body burns fewer calories at rest, which increases the risk of weight regain over time.
Microdosing does not independently protect muscle. Appetite reduction lowers calorie intake, and lower calorie intake often reduces protein intake unless nutrition is intentionally structured.
Preservation must therefore be intentional.
Menopause can alter hunger and fullness signaling at the hypothalamic level. Many women report appetite patterns that feel more persistent, less responsive to satiety cues, and more emotionally reactive.
Estrogen interacts with central appetite regulation. When estrogen declines, internal hunger feedback can become less predictable.
GLP-1 receptor activation directly engages this same pathway. At validated doses, many patients experience reduced food preoccupation and earlier satiety.
Whether microdoses produce clinically meaningful appetite regulation remains uncertain. The biological mechanism suggests it could occur, but the degree of appetite change and its long-term metabolic impact have not been defined in controlled clinical trials.
In other words, the mechanism makes microdosing biologically plausible, but reliable outcomes remain tied to dose levels that have been studied.
Calibration may therefore be biologically coherent, while the strength of the evidence remains dose-dependent.
At lower dose levels, GLP-1 medications may still influence appetite, insulin signaling, and digestion, but the metabolic effects become less predictable than they are at validated trial doses.
GLP-1 receptor agonists influence metabolism through three primary pathways: appetite modulation, glucose-dependent insulin secretion, and slowed gastric emptying. At validated doses, these mechanisms produce predictable metabolic outcomes. At lower dose levels, the same biological pathways may still activate, but the magnitude and reliability of the response remain uncertain.
Understanding that distinction is important. The biological mechanism explains why lower-dose strategies are plausible, but dose size determines how strong the effect becomes. Clinical trials establish whether that effect is reliable across large populations.
Microdosing therefore sits between plausible mechanism and proven outcomes.

In large randomized clinical trials, GLP-1 therapy begins at defined initiation thresholds before gradual titration upward.
Typical starting doses include:
These starting doses were studied in programs such as the STEP and SURMOUNT trials and were gradually increased to achieve validated metabolic outcomes.
Microdosing operates below those initiation thresholds. In off-label discussions, commonly referenced ranges include:
For a closer look at typical low-dose ranges, see our tirzepatide microdosing chart.
These lower doses have not been evaluated in randomized clinical trials for weight-loss magnitude, lean mass preservation, or HbA1c reduction.
Lower doses may still activate GLP-1 receptors, but appetite suppression tends to scale with dose intensity. Because calorie intake is strongly influenced by appetite signaling, uncertainty at the dose level translates into uncertainty in expected weight change.
When patients ask our supervising MD, Dr. Ana Lisa Carr, what separates microdosing from full therapeutic dosing, the answer usually comes down to receptor activation intensity, appetite signaling strength, and the degree of metabolic change expected at each exposure level.
GLP-1 receptor activation reduces appetite signaling in areas of the brain involved in hunger regulation, particularly the hypothalamus and brainstem. At validated doses, these receptors increase satiety and reduce food preoccupation.
Many patients describe the experience not simply as “less hunger,” but as reduced mental noise around food. Appetite regulation involves both physiological hunger and cognitive drive, which is why this change can feel noticeable even before significant weight loss occurs.
At approved weight-loss doses, caloric intake typically decreases in a consistent and measurable way. Clinical trials of GLP-1 receptor agonists have shown average reductions in daily energy intake of roughly 20–30% during early treatment phases, reflecting the combined effects of increased satiety and reduced food preoccupation.
At lower doses, receptor activation may still occur, but the strength and consistency of appetite suppression have not been defined in controlled trials.
If satiety signaling increases only modestly, calorie intake may decrease slightly rather than substantially. Because body composition changes are ultimately driven by sustained energy balance, the durability of appetite suppression becomes a major factor in long-term outcomes.
Microdosing may partially engage this pathway. Whether partial engagement produces durable metabolic change remains uncertain.
GLP-1 receptor agonists enhance insulin secretion in a glucose-dependent manner. When blood glucose rises, insulin release increases. When glucose normalizes, insulin secretion falls, which helps reduce the risk of hypoglycemia.
At validated doses, GLP-1 therapy commonly reduces HbA1c by roughly 1.0–1.5 percentage points in clinical trials. Weight loss also tends to follow a dose-response pattern, meaning higher doses generally produce greater average reductions in body fat.
Microdosing operates below the studied initiation threshold. As a result, there are currently no randomized trials showing how much glucose control or fat mass change occurs at these lower doses.
Improved insulin signaling can support metabolic stability by helping the body regulate glucose more efficiently. When glucose levels stabilize, the metabolic pressure that promotes fat storage may decrease. The open question is whether microdosing produces enough of that effect to create meaningful clinical change.
In other words, the question is not whether GLP-1 influences insulin signaling. The question is whether microdosing influences it strongly enough to produce measurable metabolic results.
GLP-1 receptor activation slows gastric emptying, meaning food leaves the stomach more slowly. Slower gastric emptying helps blunt post-meal glucose spikes and can prolong feelings of fullness after eating.
At therapeutic doses, GLP-1 medications can delay gastric emptying by roughly 30–50% during the early phases of treatment, which contributes to reduced post-meal glucose spikes and improved satiety signaling.
This mechanism likely activates even at lower doses because it is driven by receptor signaling. However, the clinical impact of partial gastric slowing without strong appetite suppression remains unclear.
When gastric emptying slows, glucose enters the bloodstream more gradually after meals. This can reduce insulin spikes and improve short-term glucose control. Over time, smoother glucose regulation may reduce some of the metabolic stress associated with repeated glucose surges.
However, without robust appetite modulation, the long-term effect on body composition may be limited.
Biological plausibility exists. Validation at microdose levels does not.
Progesterone decline can influence sleep quality, and sleep quality can influence how well GLP-1 medications are tolerated. This connection is often overlooked in discussions about metabolism during perimenopause and menopause.
Progesterone helps support calming signals in the brain that promote stable sleep. As progesterone fluctuates and gradually declines during perimenopause, sleep can become more fragmented and less restorative.
When sleep becomes disrupted, stress signaling through the brain–gut axis often increases, and sleep disturbance has been associated with worsening gastrointestinal symptoms. Higher cortisol levels are also associated with increased appetite and greater physiologic stress signaling in the body. Elevated stress signaling may increase gastrointestinal sensitivity and amplify how nausea or digestive discomfort are perceived.
Because GLP-1 medications already influence digestion and appetite regulation, poor sleep can amplify the way side effects are perceived.
Women with untreated progesterone deficiency may therefore experience stronger GLP-1 side effects regardless of dose. In those situations, lowering the dose may temporarily reduce symptoms, but it does not address the underlying sleep disruption that is contributing to the problem.
Stable sleep tends to improve medication tolerance. When medications are tolerated more comfortably, dose adjustments become easier to sustain over time, which is important for long-term metabolic outcomes.
For this reason, progesterone evaluation belongs in the Stabilize phase of the metabolic framework rather than as an afterthought during Calibrate.
Systemic estradiol can influence metabolic regulation, while vaginal estradiol primarily treats local symptoms. Understanding this difference is important when evaluating GLP-1 dosing strategies in menopause.
Systemic estradiol enters the bloodstream and circulates throughout the body. It may be delivered through patches, gels, sprays, or oral formulations. Because it circulates systemically, estradiol can influence several metabolic systems, including:
Vaginal estradiol works differently. It is designed to treat genitourinary symptoms such as vaginal dryness and discomfort. At standard doses, most of the medication remains localized to vaginal tissue and produces minimal systemic hormone exposure.
Because systemic estrogen affects insulin sensitivity and fat distribution, it can influence the broader metabolic baseline that GLP-1 therapy interacts with. When that baseline shifts, medication response can shift with it.
This distinction becomes important when discussing GLP-1 calibration.
A woman using vaginal estradiol for symptom relief may still experience many of the systemic metabolic effects of estrogen decline. In those cases, metabolic stabilization may not yet be complete.
Hormonal status influences insulin sensitivity, and insulin sensitivity strongly affects how the body responds to GLP-1 medications. For that reason, dose strategy should consider systemic hormonal status rather than symptom relief alone.
Microdosing decisions in the Calibrate phase should therefore take systemic hormone status into account, not simply whether vaginal symptoms have improved.
Lean mass preservation is essential during midlife GLP-1 therapy because estrogen decline already accelerates muscle loss. When weight reduction occurs without a plan to protect muscle, that natural decline can become more pronounced.
Clinical trials illustrate this risk. In the STEP 1 trial, approximately 38–40% of total weight lost during semaglutide treatment came from lean mass. For a woman losing 25 pounds, that could represent roughly 10 pounds of muscle if resistance training is not part of the plan.
Lean mass plays a major role in resting metabolic rate, and resting metabolic rate strongly influences weight stability over time. When muscle mass declines, the body burns fewer calories at rest, which can increase the likelihood of weight regain.
This is why the Preserve phase exists in the hormone-aware metabolic sequence.
Microdosing does not independently protect muscle. Even modest appetite reduction can unintentionally lower protein intake. Without sufficient protein and mechanical stimulus, muscle protein synthesis gradually declines.
Muscle protein synthesis depends on two primary signals:
Resistance training activates the pathways responsible for muscle repair and growth, while dietary protein provides the amino acids required for rebuilding muscle tissue.
Together, these inputs help preserve lean mass during periods of weight change.
In practical terms, preservation usually requires:
Without these inputs, weight loss may still occur, but a larger proportion of that loss may come from lean tissue rather than fat.
Because lean mass supports metabolic rate and metabolic rate supports long-term weight stability, preserving muscle becomes one of the most important factors in maintaining results.
Calibration without preservation can therefore create a metabolic tradeoff rather than a metabolic improvement.
GLP-1 microdosing is biologically plausible, but its safety and effectiveness have not been validated in randomized clinical trials. That distinction matters more than enthusiasm.
At this time, there are no randomized controlled trials evaluating the safety or effectiveness of GLP-1 microdosing protocols in any population.
Major medical organizations such as the American Diabetes Association (ADA) and the Endocrine Society publish clinical guidance on GLP-1 therapy. Their recommendations focus on treatment at validated doses that were studied in large clinical trials. Those guidelines do not define dosing strategies below the established initiation thresholds.
Academic medical centers have also noted the lack of structured data at microdose levels. Institutions including UCLA Health and Cleveland Clinic have publicly stated that outcomes at these lower doses remain poorly studied.
Biological mechanisms explain why lower-dose strategies may still produce metabolic effects. However, clinical trials are what establish whether a treatment produces reliable outcomes across large populations, and professional guidelines define the standard of care physicians follow in practice. At the moment, microdosing satisfies the first condition but not the latter two.
This does not mean microdosing never works. It means we do not yet have structured evidence defining expected fat loss, lean mass changes, long-term durability, or safety at those lower doses.
Absence of evidence is not proof of failure. It is simply a boundary of certainty.
Compounded GLP-1 medications introduce a separate layer of variability that is independent of dose size.
The FDA has issued warnings regarding certain compounded semaglutide and tirzepatide products, citing concerns related to purity, dosing accuracy, and contamination. These warnings reflect the fact that compounded medications follow a different regulatory pathway than brand-name pharmaceuticals.
Brand-name GLP-1 medications are manufactured under strict federal pharmaceutical oversight with standardized quality controls. Compounded medications are prepared by state-licensed pharmacies that operate under different regulatory frameworks.
Manufacturing standards play a role in how precisely a medication is produced. Greater precision tends to produce more predictable pharmacologic effects, while variability can introduce uncertainty in both dosing and outcomes.
Lower cost does not automatically mean higher risk. However, lower prices often reflect differences in manufacturing oversight and regulatory control.
When microdosing is combined with compounded sourcing, two uncertainties may overlap:
This does not automatically invalidate compounded medication. It simply means decisions should be informed rather than casual.
Another risk layer appears when GLP-1 medications are prescribed without full metabolic evaluation.
A short intake questionnaire cannot replace a comprehensive medical assessment that includes health history review, medication reconciliation, laboratory evaluation, and cardiovascular risk screening.
Safe GLP-1 therapy generally requires:
Lowering the dose does not eliminate contraindications. A smaller dose does not neutralize risk in patients with conditions such as medullary thyroid carcinoma history, MEN2, pancreatitis, gastroparesis, or pregnancy.
In clinical care, eligibility determines whether a patient is an appropriate candidate for treatment. Once candidacy is established, treatment structure and monitoring determine how safely therapy can be delivered. Dose size alone cannot override underlying physiology or medical risk.
Microdosing may have a role in a few supervised clinical situations. It is not intended to be a universal starting strategy.
In most cases, GLP-1 therapy begins at validated initiation doses that were studied in large clinical trials. Microdosing may be considered when standard starting doses create barriers to tolerance or continuation.
At Josie, Dr. Ana Lisa Carr will determine whether semaglutide or tirzepatide microdose fits your biological makeup.
Three scenarios may support lower initial dosing under physician supervision.
Some women experience significant gastrointestinal side effects when starting GLP-1 therapy. Beginning below the usual initiation threshold and gradually increasing the dose may help the body adjust more comfortably.
Lower initial exposure may reduce early side effects, and improved tolerability can make it easier for patients to remain on therapy long enough to evaluate its benefits.
Some patients have metabolic profiles that support GLP-1 therapy but struggle to tolerate standard starting doses. In these cases, a lower-dose approach may allow partial receptor engagement while clinicians monitor symptoms and metabolic markers.
Partial receptor activation may still influence appetite signaling, which can affect calorie intake. Because calorie intake ultimately drives changes in body composition, even modest appetite modulation could influence weight trends. However, the degree of metabolic change at these lower doses remains uncertain.
Patients transitioning off higher GLP-1 doses may benefit from gradual reduction rather than abrupt discontinuation.
Gradual dose reduction may help stabilize appetite signals during the transition. When appetite rebounds abruptly, rebound hunger can increase the likelihood of rapid weight regain. A structured taper may help reduce that risk for some patients.
In all of these scenarios, medical supervision and metabolic monitoring remain important.
Microdosing is not appropriate when core safety or metabolic foundations have not been addressed.
Examples include:
Reducing the dose does not remove contraindications or eliminate underlying medical risks. It also does not prevent muscle loss or replace the need for hormonal stabilization when systemic deficits are present.
Context determines whether a dosing strategy is appropriate, and structured medical oversight helps determine whether treatment can be delivered safely.
Decisions about GLP-1 microdosing should follow a structured evaluation rather than social media trends. The following checklist operationalizes the Stabilize → Calibrate → Accelerate → Preserve framework introduced earlier in this guide.
Each step helps determine whether dose calibration is occurring on a stable metabolic foundation.
Has systemic estradiol and progesterone status been evaluated by a menopause-informed clinician?
If systemic hormone deficits remain unaddressed, the metabolic baseline may remain unstable. Hormonal changes influence insulin sensitivity, which in turn affects how the body responds to GLP-1 medications. When hormonal conditions fluctuate, medication response may also become unpredictable.
Without hormonal stability, dose adjustments may produce inconsistent results.
Resistance training at least twice per week is the minimum structural input for protecting lean mass during weight loss.
Without mechanical stimulus, muscle loss becomes more likely during calorie reduction. Because muscle mass influences metabolic rate, and metabolic rate influences weight stability, preserving lean tissue becomes a central part of maintaining long-term results.
Strength training therefore supports the Preserve phase of the metabolic framework.
Objective data helps determine whether therapy is working and how adjustments should occur.
Baseline markers may include:
Without baseline measurements, it becomes difficult to determine whether metabolic changes are occurring or whether dose adjustments are producing meaningful effects.
Reliable data allows clinicians to identify trends, make informed adjustments, and guide treatment direction over time.
Supervision involves more than writing a prescription. Proper clinical oversight includes review of medical history, medication interactions, cardiovascular risk, and ongoing monitoring.
When dose adjustments occur without monitoring, uncertainty increases and potential risks may go unnoticed.
Clinical supervision supports safe titration, and consistent titration practices help maintain treatment stability over time.
Microdosing strategies have not been validated in randomized controlled trials.
If confidence in this approach depends on the belief that it has already been studied at scale, that belief is not supported by current evidence.
Understanding these limitations helps set realistic expectations and encourages more cautious decision-making, which ultimately reduces risk.
Lower risk generally supports greater long-term sustainability.
The hormone-aware metabolic framework places GLP-1 microdosing within a broader system of metabolic care rather than treating it as a stand-alone solution.
Within this framework, microdosing functions as a form of calibration. It is not a method of stabilizing hormones, and it does not replace the need to preserve lean mass during weight loss.
Hormonal stabilization typically comes first. When systemic estrogen or progesterone deficits remain unaddressed, the metabolic baseline may remain unstable, and medication responses can become less predictable.
Strength training plays a separate but equally important role. GLP-1 medications influence appetite and glucose regulation, but they do not independently preserve muscle mass. Resistance training and adequate protein intake remain the primary tools for maintaining lean tissue during weight reduction.
Escalation decisions should rely on objective data rather than subjective tolerance alone. Metabolic markers, body composition trends, and clinical monitoring help determine whether a dosing strategy is producing durable results.
Hormonal systems also interact with medication tolerance in ways that are often overlooked. Progesterone influences sleep quality, sleep disruption can increase cortisol levels, and elevated cortisol may amplify side effects such as nausea or digestive discomfort.
Hormone type also matters. Vaginal estradiol primarily treats local genitourinary symptoms, while systemic estradiol can influence broader metabolic processes such as insulin sensitivity and fat distribution.
When these elements are addressed in the correct order, GLP-1 therapy becomes part of a structured metabolic strategy rather than an isolated intervention. In midlife metabolic care, sequence often determines long-term durability.
GLP-1 microdosing sits between biological plausibility and clinical uncertainty. While the mechanisms of GLP-1 medications are well understood, reliable outcomes below validated dosing thresholds have not been established in clinical trials.
For women in perimenopause and menopause, dosing decisions should follow a structured evaluation. Hormonal stability, lean mass preservation through strength training, and baseline metabolic data all shape how the body responds to medication.
When these factors are addressed first, GLP-1 therapy becomes part of a coordinated metabolic strategy rather than a stand-alone solution. In midlife care, the sequence of intervention often determines the sustainability of results.
GLP-1 microdosing should never be self-directed or trend-driven. If used, it should follow hormonal evaluation, metabolic lab review, and a lean mass preservation strategy.
For women seeking a menopause-specific clinical pathway, structured programs are available that incorporate:
If you are exploring clinician-guided microdosing within a defined menopause framework, you can review available compounded GLP-1 calibration options below.
These options are intended for supervised use within a structured care model.
Month-to-month:
3-Month Bundle and Save:
Frontiers in Endocrinology.
GLP-1 Physiology and Metabolic Regulation.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12476180/
Frontiers in Endocrinology.
GLP-1 Receptor Agonists and Cardiometabolic Effects.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11620716/
Cells.
GLP-1 Receptor Biology and Metabolic Signaling.
https://www.mdpi.com/2073-4409/13/1/65
American Journal of Obstetrics and Gynecology.
Hormonal Regulation of Appetite and Metabolism in Women.
https://www.sciencedirect.com/science/article/pii/S0002934325000592
Jastreboff AM, Aronne LJ, Ahmad NN, et al.
Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1).
New England Journal of Medicine.
https://www.nejm.org/doi/full/10.1056/NEJMoa2206038
Khanijow V, Prakash P, Emsellem HA, et al.
Sleep Dysfunction and Gastrointestinal Diseases.
Gastroenterology & Hepatology.
https://pubmed.ncbi.nlm.nih.gov/27134599/
Mauvais-Jarvis F.
Sex Differences in Metabolic Regulation and Insulin Sensitivity.
Biology of Sex Differences.
https://pmc.ncbi.nlm.nih.gov/articles/PMC10504839/
Morton RW, Murphy KT, McKellar SR, et al.
A Systematic Review of Protein Intake and Muscle Mass in Resistance Training.
British Journal of Sports Medicine.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4354766/
Santollo J, Daniels D.
Control of Appetite and Energy Balance by Estrogens.
Physiology & Behavior.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6341301/
Schüssler P, Kluge M, Yassouridis A, et al.
Progesterone Reduces Wakefulness in Sleep EEG Studies.
Psychoneuroendocrinology.
https://pubmed.ncbi.nlm.nih.gov/17168724/
Toth MJ, Tchernof A, Sites CK, Poehlman ET.
Menopause-Related Changes in Body Fat Distribution.
International Journal of Obesity.
https://pubmed.ncbi.nlm.nih.gov/10865795/
Wilding JPH, Batterham RL, Calanna S, et al.
Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP-1).
New England Journal of Medicine.
https://www.nejm.org/doi/full/10.1056/NEJMoa2032183
Frontiers in Neuroscience.
Sleep Disturbance and Gastrointestinal Symptom Sensitivity: Brain–Gut Axis Mechanisms.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11340591/